White Papers, Case Studies, Reports


From three meetings to one removing bottlenecks with AI-enabled eCOA
Discover how AI-enabled eCOA and agentic workflows reduce clinical trial startup time, translation cycles, and meeting overhead—cutting eCOA build timelines from 16–20 weeks to under 8 weeks.


Report: Medable named #1 Leader in DCT for second year by Everest Group
For the second consecutive year, Medable has been named Leader decentralized clinical trial products per the Everest Group’s PEAK Matrix® assessment, which evaluates DCT products from 24 companies based on vision, capability and market impact.


ePRO Case study: Better options and outcomes oncology trials.
Medable worked with a top 5 pharma company to increase the safety of patients as anti-cancer treatments may cause pulmonary toxicity, ranging from asymptomatic radiological changes to respiratory failure, and is considered a common side effect.
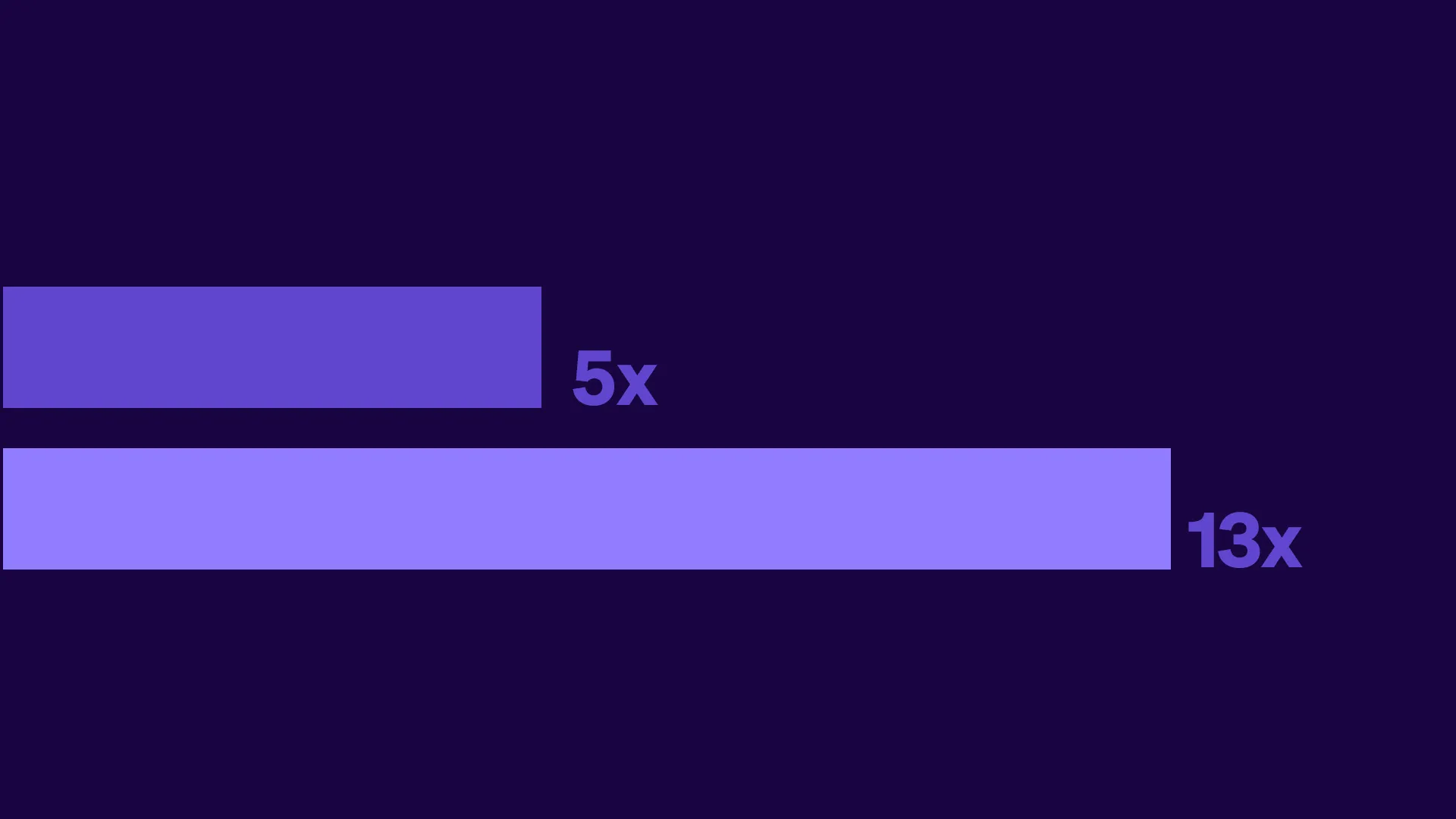

Tufts CSDD Impact report: Decentralized and hybrid trials deliver greater ROI than traditional trials
The Tufts Center for the Study of Drug Development completed an analysis of its financial modeling study of decentralized clinical trials using real data from Medable-enabled studies. These findings net financial benefits ranging from 5x for Phase II and 13x for Phase III trials, equating to roughly $10 million ROI and $39 million ROI, respectively.


White paper: Why eConsent primes patients and studies for success
Read this white paper to learn how eConsent's flexibility helps you educate and retain patients worldwide, regardless of signature requirements, and how to implement it across your organization.


White paper: Tips for tailoring eConsent for optimal patient centricity.
Use a consent experience that’s designed to meet the needs of patients, while educating and engaging them each step of the way. Read our free white paper to learn eConsent tips from patients themselves, and improve your patient retention.


White paper: The foundations of decentralized clinical trials
Understand the benefits of web-enabled research tools and how they work.

White paper: The building blocks of DCT. How to create a seamless experience across eConsent, eCOA and more
Combining multiple market solutions at the site level can result in a negative experience due to different processes, login credentials, and redundant training. A unified DCT platform that works seamlessly with other core clinical systems to streamline workflows and simplify key trial processes.




