White Papers, Case Studies, Reports


From three meetings to one removing bottlenecks with AI-enabled eCOA
Discover how AI-enabled eCOA and agentic workflows reduce clinical trial startup time, translation cycles, and meeting overhead—cutting eCOA build timelines from 16–20 weeks to under 8 weeks.


Case study: Scaling global vaccine mega-trials for a top-5 pharma
Learn how Medable enabled a top-5 pharma to scale vaccine mega-trials with near-100% enrollment, real-time safety data, and >90% diary compliance.


Case study: ICON and Medable drive 85% eConsent adoption in U.S. menopause study
How do you drive adoption in a complex women’s health study? See how ICON and Medable reached 85% eConsent uptake across 1,200+ participants with a smarter, site-first approach.


Medable oncology solutions
Discover how Medable’s AI-powered oncology platform simplifies complex cancer trials by integrating eCOA, ePRO, and eConsent solutions—reducing trial time, improving patient retention, and enhancing data quality for faster, more efficient research.


Case study: Driving a high-adherence LTFU trial without an EDC
Learn how Medable is powering a decade-long, global long-term follow-up (LTFU) obesity study, achieving an impressive 97% patient retention rate without using a traditional EDC system all while delivering a compliant, scalable, and cost-efficient solution.


Eliminate clinical trial white space with the right AI strategy
It has become clear that our industry has reachedthe limits of human-only clinical development. As clinical trials have become increasingly complex, the endeavors that people alone can perform are no longer sufficient to generate the momentum needed to address the growing burden of human disease. This has led to longer drug development timelines and significant delays for patients. One large are of lost time is “white space,” definied simply as unproductive time caused by manual, sequential processes and fragmented data systems. Thankfully, a solution lies in agentic AI and its abilities to perform series of tasks.


Roadmap to adopting AI agents
The successful integration of AI agents in enterprise operations requires a balanced, deliberate approach. Drawing from recent research in Strategic Integration (SI) and agentic AI adoption within large enterprises, the following best practices help maximize value, manage change effectively, and mitigate common pitfalls. Medable Agent Studio specifically streamlines this process by providing robust tools, no-code simplicity, and built-in compliance and security standards.


Guidance for using AI agents effectively
AI agents can play a range of roles, from acting as intelligent collaborators alongside people to independently executing entire workflows. Rooted in the principles of agentic AI, they excel at reasoning through problems, self-directing activities, and adapting to evolving contexts. Whether deployed to support individuals or to autonomously carry out operational tasks, agents enhance productivity, decision-making, and overall efficiency across a broad spectrum of use cases.


An overview of Agent Studio
Agent Studio reimagines the clinical trial lifecycle, automating slow, manual processes, removing barriers in the clinical process, and introducing new ways to achieve clinical goals like never before. This first-of-its-kind, AI-powered, no-code platform lets you deploy ready-to-go agents trained as clinical development experts or create bespoke ones using your own data and expertise, unlocking endless possibilities.


Rewriting the Future of Clinical Trials: AI, Agility, and the Portfolio-First Mandate
In a rapidly changing research landscape, leaders at Takeda, Novartis, Sanofi, Daiichi Sankyo, and Medable share firsthand insights into how artificial intelligence is reshaping clinical trial operations.
This exclusive whitepaper explores how forward-thinking organizations are reimagining trial design, execution, and scaling to meet the demands of speed, precision, and patient-centricity across their entire portfolio.
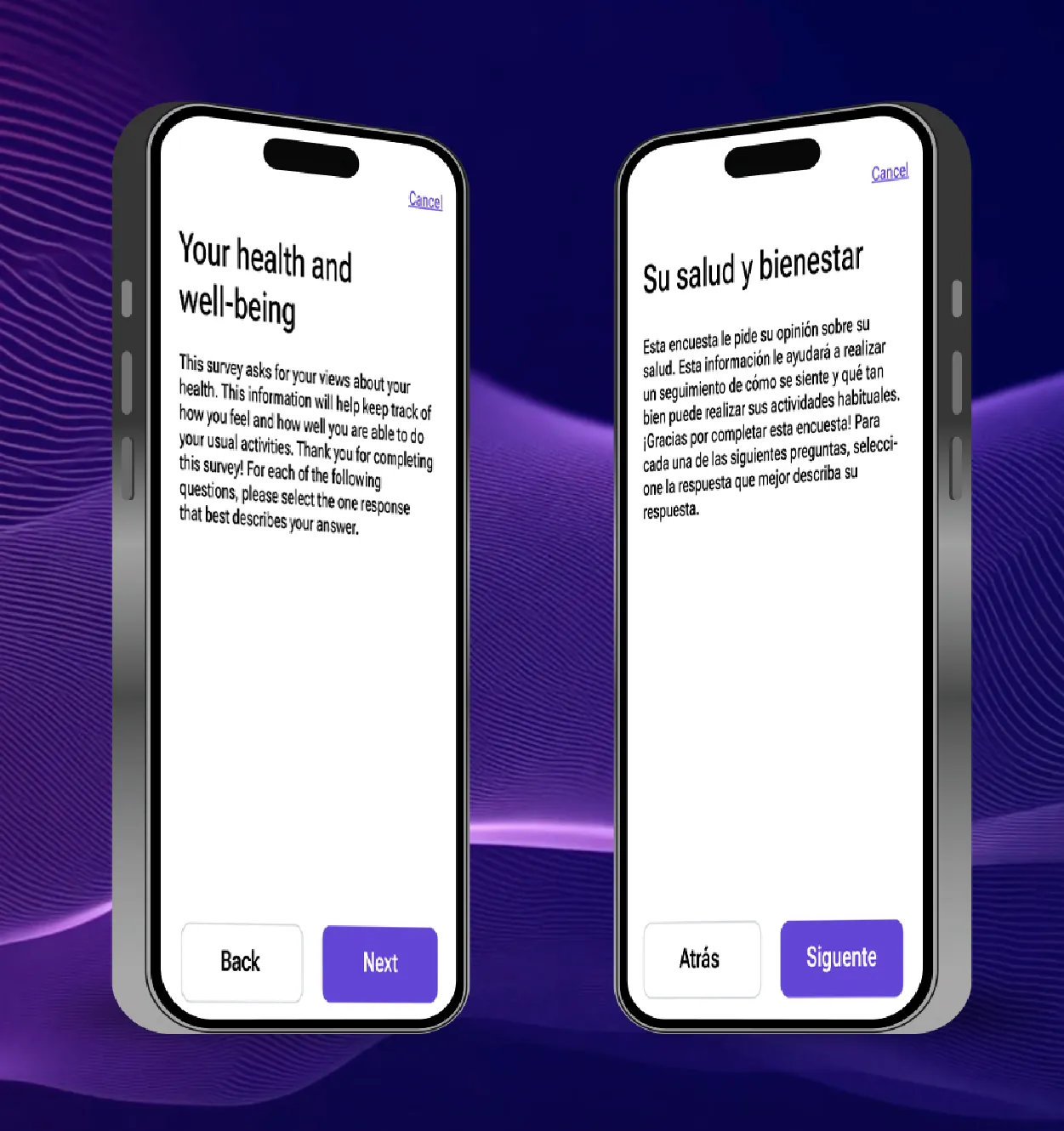

Case study: Removing translation bottlenecks with AI
Traditionally, translations and language migration create significant bottlenecks on the path to trial study go-live. The process is traditionally manual, linear, and resource intensive. To address these challenges, Medable partnered with Lionbridge, a leading translation services company to compare the status quo translation process against an AI-enabled approach powered by both companies’ proprietary new AI tools.


Case Study: Medable, CRO, & pharma partner to accelerate clinical trial deployment at scale
A top-3 contract research organization (CRO) and a top-10 pharmaceutical company partnered with Medable to help accelerate the speed, scale, and standardization of their eCOA trials.
The two organizations had been hampered by lengthy configuration timelines, inconsistent designs across studies, and siloed processes between sponsors and their CRO partners.
By centralizing the sponsor’s preferred instruments and configurations into a reusable digital library, Medable enabled the CRO to rapidly accelerate its portfolio development and initiate new studies without starting them “from scratch.”
Complete the form to get your copy of the case study and see how we enable rapid acceleration for CROs.


Accelerating oncology research: Digital strategies for modern clinical trials
Learn how digital tools ease oncology research for all stakeholders while accelerating the pace of research with this whitepaper.




