Your co-pilot for clinical monitoring
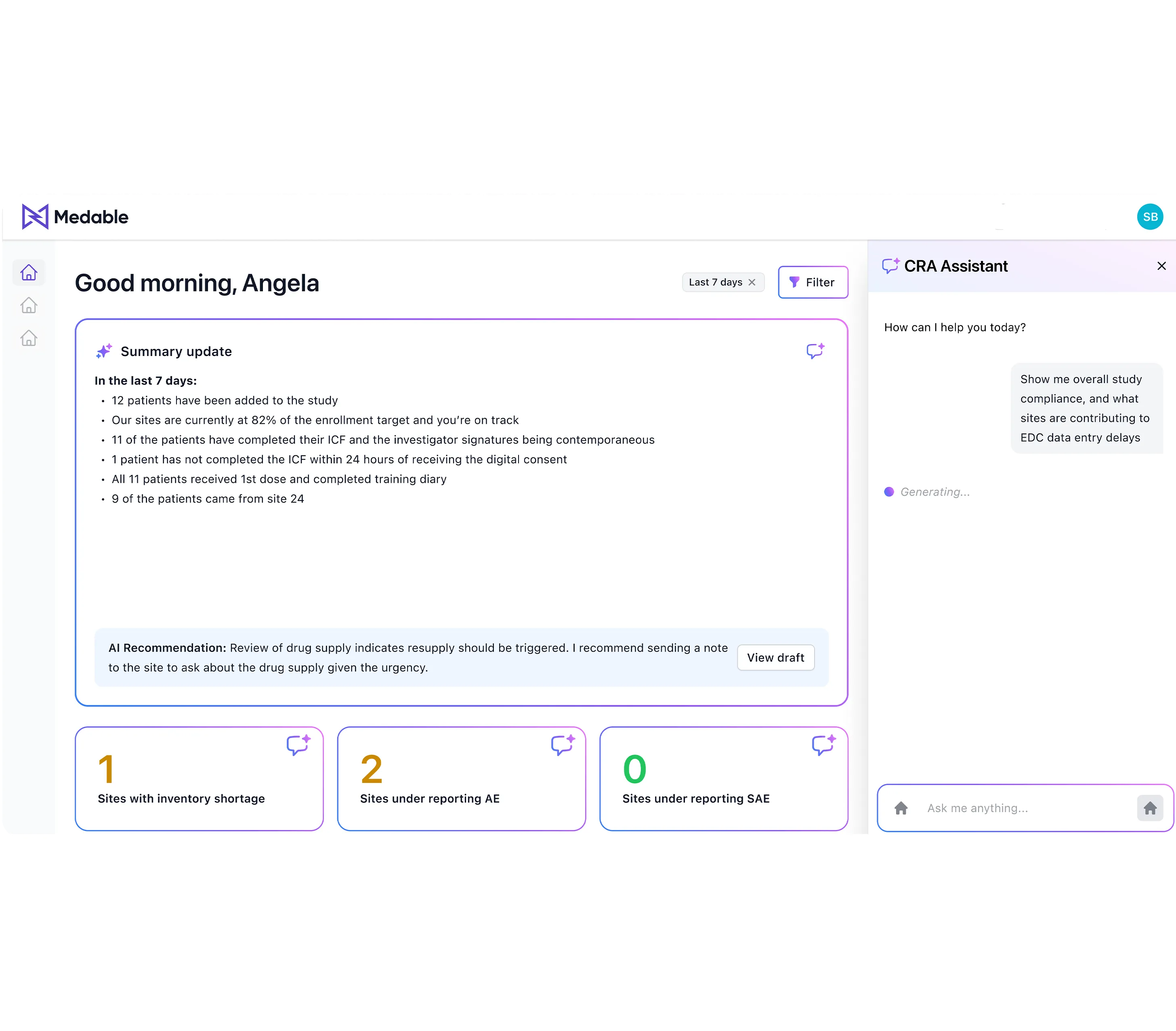
Medable’s CRA Agent is an AI-driven solution that automates and optimizes clinical trial monitoring by proactively identifying and prioritizing site risks, generating comprehensive pre-visit summaries, and providing actionable recommendations to enhance trial oversight and compliance.

We estimate that CRA agents can take on up to 90% of the tactical and administrative work the CRA handles on a daily basis — from sending site-specific emails and reminders to tracking responses and updating systems. That means we can focus our time on the strategic decisions that move trials forward, while standardizing and streamlining the tasks that used to consume most of our day.

Connected monitoring across systems
Perceive signals across the entire clinical data ecosystem by unifying CTMS, RTSM, EDC, labs, consent, and safety into one view. Turn manual checks into automated insights, instantly highlighting which sites are on track and where intervention is needed, saving valuable time, system-hopping, and manual data compilation.

Human-in-the-loop oversight
Easily identify risks and recommendations for best-next actions tied to your protocol, consent, and regulatory requirements. Stay assured with a transparent reasoning trail thatshows why each step is advised, enabling CRAs to collaborate confidently with sites, improve decision-making, enhance their compliance, all while freeing CRAs to focus on engagement and site-specific challenges.
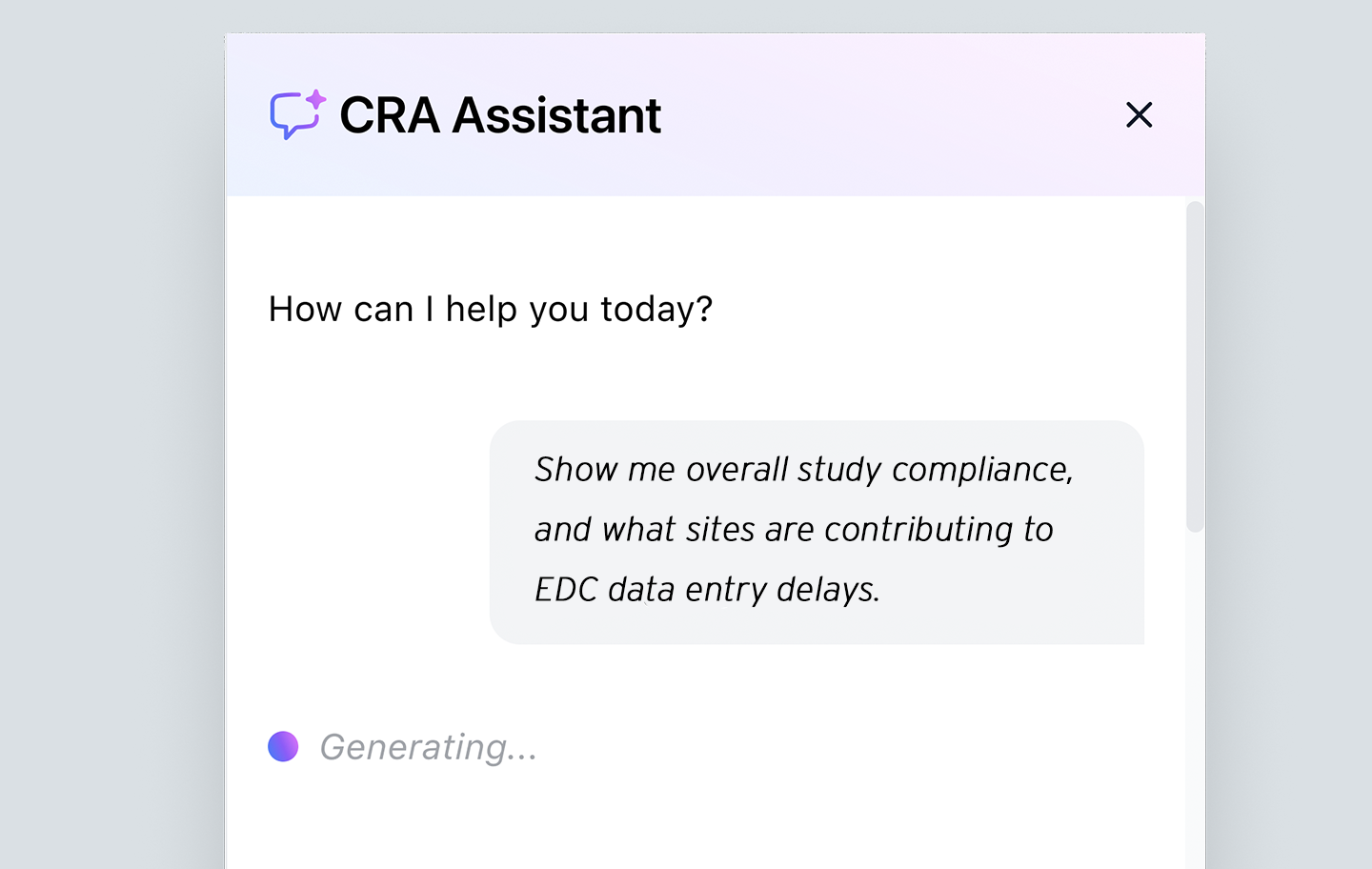
Automated administrative tasks
Take advantage of automated routine tasks like drafting queries, sending emails, and updating CTMS/eTMF — always with human CRA oversight. Close the loop between planning and execution with real-time tracking that reduces administrative burden, ensures data consistency, and accelerates trial progress.
The latest from Knowledge Centers


Real-time clinical trials are here. Medable was built for them.
Real-time clinical trials are here. Medable's continuous trial management platform gives sponsors the evidence generation, agentic AI, and regulatory-ready infrastructure to run clinical programs at the speed the FDA now expects.


Key criteria for evaluating AI and agentic AI clinical trial vendors
Artificial Intelligence is rapidly transforming clinical research. From patient recruitment and protocol design to medical writing and data review, AI-powered solutions are becoming embedded across the clinical development lifecycle. More recently, the emergence of Agentic AI (systems capable of planning, reasoning, and executing multi-step workflows with varying degrees of autonomy) has generated significant excitement throughout the industry.
However, not all AI solutions are created equal. While many vendors promise dramatic improvements in efficiency and productivity, clinical trial organizations operate in one of the most highly regulated environments in the world. Success depends not only on technical performance but also on compliance, validation, governance, security, and trust.
As sponsors, CROs, and technology teams evaluate potential AI partners, they need a framework that extends beyond traditional software procurement criteria. The following considerations can help organizations assess both AI and Agentic AI vendors and identify solutions that are truly ready for clinical research.


Building a scalable oncology engine in a dynamic market
Learn how a a top-10 global pharmaceutical company partnered with Medable to rapidly expand the number of oncology trials it could concurrently conduct.

Eliminate clinical trial white space with the right AI strategy
It has become clear that our industry has reachedthe limits of human-only clinical development. As clinical trials have become increasingly complex, the endeavors that people alone can perform are no longer sufficient to generate the momentum needed to address the growing burden of human disease. This has led to longer drug development timelines and significant delays for patients. One large are of lost time is “white space,” definied simply as unproductive time caused by manual, sequential processes and fragmented data systems. Thankfully, a solution lies in agentic AI and its abilities to perform series of tasks.




