
We’re on a mission to enable effective therapies to people faster.
A novel approach to research
For the past two decades clinical research has been hindered by outdated processes and technologies leaving millions without treatments.
After struggling to access patients in her own research in rare skin diseases, Dr. Michelle Longmire set out to solve this long-standing challenge in research.
Drawing inspiration from innovation and technological advancements in other industries, Longmire rallied stakeholders, partners, and developers to launch our company, Medable, with the shared vision of radically increasing the number of effective therapies to people each year.
Committed to science. Powered by innovation.
In order to achieve this vision, we needed to amplify the quality, reach, velocity, and cost of research.
Driven by the belief that clinical research can and should be better, we've assembled world-class scientific and product teams to continuously drive evidence-based improvements to the trial experience for all.
The relentless pursuit of progress, together.
Human connection is at the heart of everything we do. We prioritize the personal experience of patients, caregivers, knowing that their knowledge and feedback are essential to improving clinical research. In close partnership with with our network of sites, we continuously improve our platform, because we know that when they win, we all win. By designing products that are more accessible and user-friendly, we can bring life-change treatments to those who need them faster.
Together, with our clients and partners, we continue to work toward our mission and drive clinical research forward for the betterment of all.
Accolades





The latest from Medable


Eliminate clinical trial white space with the right AI strategy
It has become clear that our industry has reachedthe limits of human-only clinical development. As clinical trials have become increasingly complex, the endeavors that people alone can perform are no longer sufficient to generate the momentum needed to address the growing burden of human disease. This has led to longer drug development timelines and significant delays for patients. One large are of lost time is “white space,” definied simply as unproductive time caused by manual, sequential processes and fragmented data systems. Thankfully, a solution lies in agentic AI and its abilities to perform series of tasks.


More than a tool: How AI agents are transforming clinical trials |
Explore how agentic AI is transforming clinical trials by automating routine tasks and boosting team efficiency with real-world, low-risk use cases.


Why cloud-based eCOA software is becoming the industry standard
The landscape of clinical trials is evolving rapidly, and at the forefront of this transformation is eCOA (electronic Clinical Outcome Assessment) software. This technology is streamlining data collection, enhancing patient compliance, and ensuring regulatory adherence like never before.


Medable Launches Agentic Accelerator Program to Help Life Sciences Companies Deploy Agentic AI Across Clinical Development Lifecycle
High-touch enablement helps sponsors operationalize industry-specific agents to realize value faster.
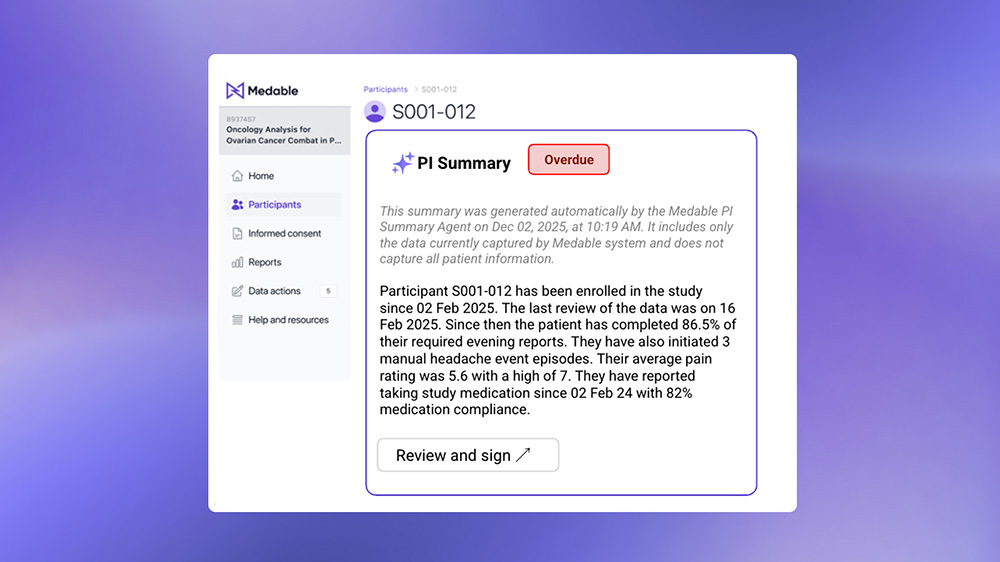

Medable Introduces AI Agent to Reduce Burden at Research Sites by Assisting Principal Investigators with Oversight of eCOA Data
Medable Inc., the leading technology platform for AI-powered clinical development, today announced the launch of Agentic AI for research sites to reduce burden and assist principal investigators in oversight and monitoring of eCOA data.


Medable Debuts AI Agent for Automating Trial Master File (TMF) Processes
TMF Agent significantly improves quality and consistency while drastically reducing the manual effort in document management. It is built on Medable’s Agent Studio, the industry’s first agentic AI platform for clinical development.

