Blog posts


The new blueprint for oncology trials: agility, consistency, and scale
Oncology has never been in a better scientific position. Precision medicines, adaptive study designs, and biomarker-driven cohorts have opened up treatment possibilities that simply did not exist ten years ago. But the complexity of running these trials has grown at much the same pace as the science itself, and that gap between scientific ambition and operational capability is where many programmes quietly struggle.
For sponsors building oncology portfolios, and for the CROs executing them, the operational challenge is no longer a peripheral concern. Getting it right comes down to three things: being consistent enough to build efficiently, agile enough to adapt when the science changes, and scalable enough to manage a growing portfolio without the overhead growing at the same rate.
Medable has worked with sponsors and CROs across many global oncology programmes, spanning thousands of sites and participants. That experience has given us a clear picture of what separates programmes that move well from those that get stuck.


Playing catch-up: FDA wants “patient’s voice” ePRO in your oncology trial
For years now, the FDA has been making one point crystal clear to sponsors and CROs across our industry; they want the patient voice incorporated whenever possible in oncology trials.
The FDA's initiative is driven by the recognition that a patient's personal experience with a disease and its treatment is a unique and essential measure of a medical product's benefit and risk.
- Rationale: The FDA explicitly states that "patients provide a unique perspective on treatment effectiveness" and "some treatment effects are known only to the patient." Outcomes that truly matter to patients, such as functioning, quality of life, and the burden of side effects, are often best measured directly by the patient.
- Mandate: The Patient-Focused Drug Development (PFDD) effort, codified in part by the 21st Century Cures Act, requires the inclusion of such patient experience data in clinical research.
- Guidance series: To formalize this approach, the FDA has released a series of methodological guidance documents (the PFDD Guidance Series) that outline how stakeholders should collect, submit, and use patient input to inform medical product development.


Build vs buy: A guide on adopting AI agents for life sciences
“Big corporations can’t rely on their internal speed to match the transformation that is happening in the world. As soon as I know a competitor has decided to build something itself, I know it has lost.”
These candid sentences from Sanofi CEO, showcase one of the most common questions that’s at the forefront of every pharmaceutical company’s mind; whether to build or buy your way into the agentic and generative AI revolutions.
In life sciences, many teams start with the same instinct. They see a capable large language model, stand up a proof of concept, and feel close to a breakthrough. For most of us, AI prototypes can look magical. A chatbot summarizes visit reports, drafts emails, or answers protocol questions in minutes. The experience is so strong that teams assume production is a short step away.
Unfortunately, the gap is much bigger than it looks.
According to a recent MIT study, 95% of AI pilots will fail, as they note that “Only 5% of custom GenAI tools survive the pilot-to-production cliff, while generic chatbots hit 83% adoption for trivial tasks but stall the moment workflows demand context and customization.”
Like MIT’s example shows, moving from prototype to production in clinical research means building something validated, compliant, scalable, and integrated into real workflows. That takes far more than clever prompts. It requires domain grounding, continuous monitoring, retraining loops, robust tool orchestration, and evidence that the system is safe and auditable under regulations like GxP, HIPAA, and 21 CFR Part 11.
Many organizations only discover the hidden costs after they have committed. Internal teams often invest for two years, spend millions in sunk cost, and still never reach a dependable clinical grade system. The illusion comes from how easy it is to get an early demo working, and how hard it is to make that demo survive contact with trial reality.


Shaping intelligence: How a “human in the loop” keeps AI anchored
It’s been said that the only constant in the world is change.
For decades, clinical trials have been a human only endeavor, with teams of clinicians, study teams, and patients working hand in hand to bring the latest molecules to market. Now, a new central actor has entered the clinical paradigm, agentic artificial intelligence.
Only three years after OpenAI kicked off the artificial intelligence arms race, AI has gone from requiring users to prompt it, to pre-emptively identifying bottlenecks, safety risks, and more, thanks to agentic AI.
Agentic AI is an autonomous, goal-oriented system that uses reasoning and external tools to independently plan, execute, and adapt multi-step actions with minimal human intervention to achieve complex objectives.
Sponsors and CROs have begun using AI agents across their workforces to improve trials in ways that humans have traditionally struggled to accomplish. For instance, organizations have been creating AI agents to analyze prior trial protocols, benefiting from lessons learned across prior trials and real-world outcomes, enabling teams to anticipate risks and automating elements of submission drafting. Anomaly detection has helped teams better identify outliers in operational metrics or safety signals, prompting early interventions. Document intelligence accelerates medical writing by grounding generative outputs in verified data, which reduces cycle time without sacrificing accuracy.
However, in each of these use cases, humans remain squarely “in the loop.” Or rather, the decision making isn’t left entirely to AI. Instead, the objective is to augment clinical, regulatory, and legal teams with tools that surface the right information at the right time.
This core concept, keeping a human in the loop, is essential to clinical decision making and operations as agentic AIs, while powerful, are not inherently suited to fully autonomous operation in all regulated contexts.


What happened at ESMO AI & Digital 2025
The 2025 ESMO AI & Digital Oncology Congress, held in Berlin from November 12 to 14, highlighted the accelerating role of artificial intelligence across the oncology care continuum. Although imaging and pathology remain the most established fields for AI adoption, this year’s programming revealed a decisive shift toward workflow-integrated AI that enhances clinical operations, supports trial efficiency, and addresses the realities of patient monitoring.
Across three days of sessions, side-room conversations, and industry demonstrations, one theme was clear. AI is evolving from experimental add-on technology into a practical clinical teammate, but scaling its impact will require robust validation, seamless integration, and a sustained focus on clinician trust.


Vertical vs. horizontal: Why your Agentic AI should be built by clinical experts in life sciences
By now, most people are familiar with ChatGPT and other general-purpose AI tools such as Perplexity. These horizontal AI systems have become commonplace, assisting users with tasks like composing emails, summarizing information, or generating creative ideas. Their adaptability across a wide range of domains demonstrates the power of general intelligence and transfer learning.
However, in the high-stakes and highly regulated context of life sciences, it is important to ask whether such tools are suitable for clinical use. In this setting, a vertical agentic AI approach—purpose-built for specific domains—provides the precision, reliability, and compliance necessary for success. While both horizontal and vertical agentic AI play essential roles in the broader evolution of intelligent systems, it is the vertical approach that ensures the rigor and accountability demanded by life sciences applications.


Sponsors talk AI: Sanofi's take on the evolving role of AI in clinical trials
Artificial intelligence continues to move from experimentation to execution across the life sciences industry. On a recent episode of the AI in Business podcast, Matthew Peruhakal, Global Head of Data Architecture, Utilization, and AI Engineering at Sanofi, offered a deep look at how the pharmaceutical giant is integrating AI to transform clinical trials. From intelligent data workflows to proactive risk detection and regulatory alignment, Peruhakal described an organization reshaping its research and development operations around a new digital core.
His message was clear: AI cannot remain a side project. To make a meaningful impact, it must be embedded as a strategic capability that connects people, systems, and data across the enterprise.


Sponsors talk AI: Novartis’ take on the evolving role of AI in clinical trials
Artificial intelligence is shifting from promise to practice in biopharma, and few domains feel the impact more than clinical development. In a recent conversation on the AI in Business podcast, Zhong Lu, director of data science and AI at Novartis, outlined how AI is evolving the clinical trials process end to end. His perspective centered on building internal platforms that unite structured and unstructured data, deploying document intelligence to remove process friction, and using responsible generative AI to support scientific decision making without compromising compliance. While the regulatory bar remains high and patient trust is paramount, Novartis’s approach shows how targeted applications of AI can streamline data operations, strengthen regulatory readiness, and make both traditional and decentralized trials faster and more resilient.


Sponsors talk AI: Takeda’s take on the evolving role of AI in clinical trials
Artificial intelligence continues to influence nearly every industry, and life sciences are no exception. In a recent conversation on the AI and Business podcast, Damien Nero, Head of Data Science in US Medical at Takeda Pharmaceuticals, shared his perspective on how AI is changing the clinical trial landscape. With over 15 years of experience applying machine learning and real-world data to drug development, Nero outlined both the progress already being made and the challenges that still stand in the way of broader transformation. His insights highlight how pharmaceutical leaders can think strategically about deploying AI to balance innovation with operational efficiency.


Reclaiming your time: How sponsors can save thousands of hours with agentic AI
“For almost a decade, it’s taken nearly eight months on average to get from site identification to study startup completion, when all sites are initiated and ready to enroll patients.”
While it’s often hard to quantify the average time spent on clinical trial activities like study startup, this 2018 quote from Tufts Research author Mary Jo Lamberti showcases the very real problem regarding the time it takes to reach key trial milestones.
In fact, in the seven years since that quote, the clinical trial landscape has only become more complex, with clinical trial cycle times increasing despite technology advances. Cycle times are defined as the total duration from the approval of a clinical trial protocol to the database lock (DBL). As reported by Statista, the average clinical trial cycle from 2020 to 2024 increased by seven months.
Additionally, IQVIA recently released research indicating that almost 50% of drug development time is attributed to non-scientific delays, aka operational bottlenecks that create unnecessary gaps between critical milestones.


Six steps to help you choose the right clinical trial partner
“This year, we’ve decided to stick to paper. We know what we’re getting and we’ve always done it this way.”
That was the unwavering response from a clinical operations lead at a pioneering biotech when asked why they still relied on paper diaries for patient-reported outcomes.
Despite the growing complexity of global trials, the promise of real-time data, and the surge of digital capabilities available today, some organizations have held tight to a method of clinical trial conduct that’s increasingly as outclassed as it is outdated.
It’s well known that individuals and organizations believe that change can be daunting, stressful, and difficult, especially when the old way is familiar and entrenched. However, much like anything else, having a partner who can help guide you through the process is massively important. But, how do you find the right partner for your trials?
Before locking in a vendor, organizations must engage in a deliberate process to assess their needs, align stakeholders, and set the foundation for long-term success. This blog explores the critical steps sponsors should take before selecting a digital partner, using insights and frameworks drawn from Medable’s therapeutic area standards and industry best practices.
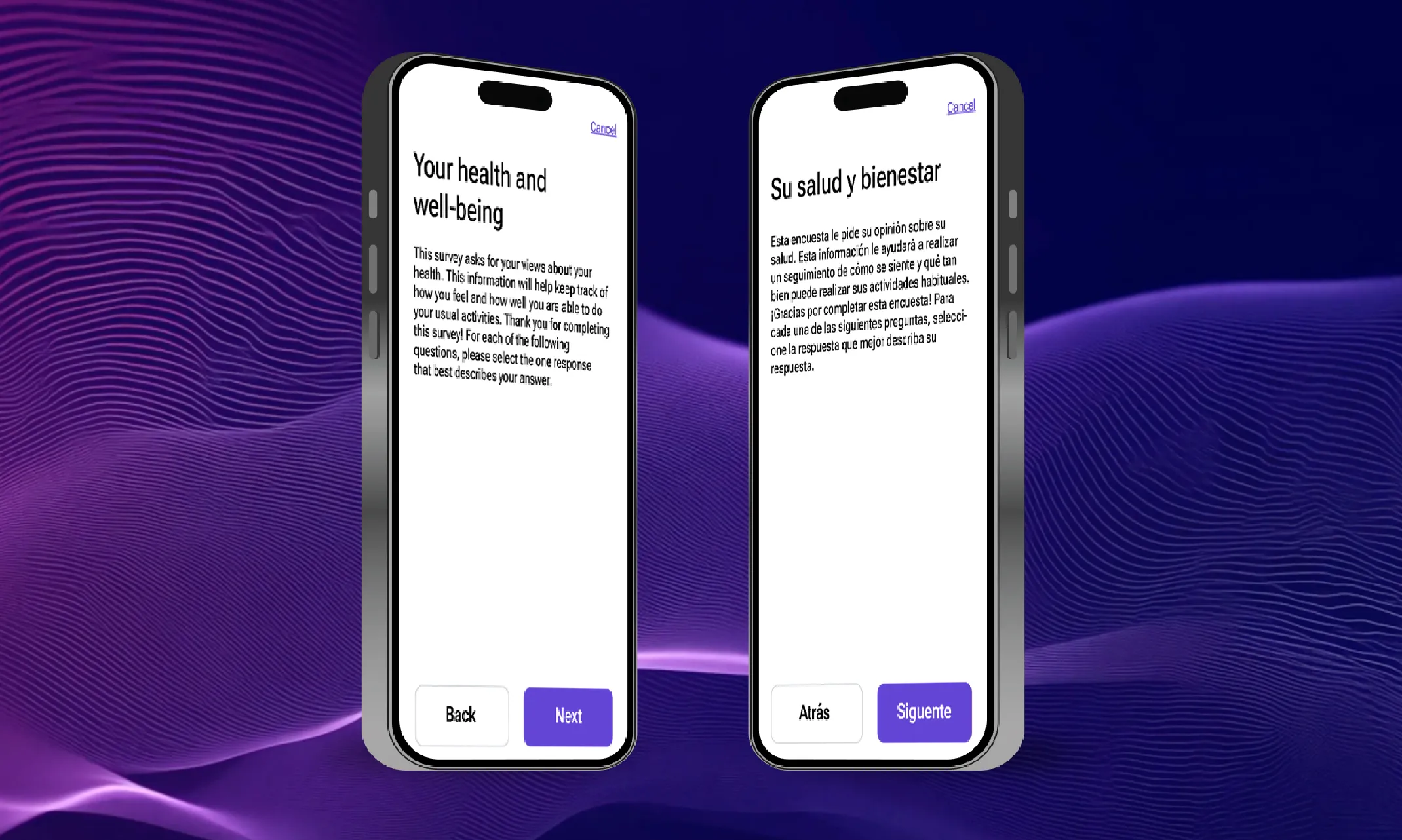

From bottlenecks to breakthroughs: How AI is transforming translation timelines
According to ClinicalTrials.gov, there are 3,046 multi-country trials being conducted this year. While many trials remain localized within a single country, there has been a definitive movement towards conducting trials in multiple countries, especially for larger, later-stage trials. This is driven by the positives that multi-country trials offer, like faster patient recruitment, lower costs in some regions, and the need for diverse patient populations.. However, behind the scenes, a critical bottleneck has been slowing many trials down. This bottleneck is the translation process that’s required to make trials work across multiple languages, locales, and regulatory bodies/organizations.


The Top 5 myths about eCOA in 2025
With MarketsandMarkets projecting the global electronic Clinical Outcome Assessments (eCOA) solutions market to grow at compound annual growth rate (CAGR) of 16.1% each year until 2030, it’s clear the eCOA has made its impact within the clinical research landscape.
Yet despite growing adoption, electronic Clinical Outcome Assessments (eCOA) are still surrounded by misconceptions that hinder their full potential. From concerns about patient usability to assumptions about cost and implementation timelines, these myths can create hesitation among sponsors and research teams alike.
Thus, we’re here to cut through the noise and set the record straight with a summary of a recent webinar featuring speakers from Transcelerate and Medable.




