Get direct access to clinical trial and patient experts
Designing a clinical trial? Looking for the latest advancements?
Get free, personalized advice from our clinical trial experts on:
- Therapeutic areas including oncology, weight loss/diabetes, vaccine, and more
- Patient experience for better data
- Protocol design and digital standardization
- New FDA & EU guidelines and how this will impact your study
- Use cases on leveraging digital for cost and time savings

“Medable is delivering quantum leap capabilities in areas of unmet need in clinical trials.”
Connect one-on-one with an expert
Designing a clinical trial? Have questions about the latest technology or keeping patients engaged?
Get free, personalized advice from our clinical trial veterans on anything from protocol design to patient experience.

Andrew MacKinnon
Executive General Manager, Customer Value
Mackinnon drives the development and application of eConsent for efficient, high-quality decentralized clinical trials. With 20 years of experience in clinical trial management across Pharma, Biotech, and CROs, he's committed to leveraging decentralized methodologies to ease clinical research burdens. Previously, he's held senior roles at Covance and focused on enhancing business performance.

Jena Daniels
VP, Customer Value - Site and Patient Engagement
Jena, renowned for her work in digital clinical trials and recognized by PharmaVoice100 and as the Best Decentralized Clinical Trial Businesswoman of 2022, pioneered Medable's Patient Success department and Patient Caregiver Network. Daniels is dedicated to ensuring patients’ lived experiences inform the clinical trial experience.

Pam Tenaerts
Chief Scientific Officer
Dr. Tenaerts is one of the leading advocates for innovation in clinical trials, with a unique personal background as a clinical trials investigator, hospital research administrator and academic in both the U.S. and Europe. Dr. Tenaerts joins Medable from Duke, where she led CTTI’s efforts to develop and drive adoption of practices that increase the quality and efficiency of clinical trials.
The latest from Medable


Eliminate clinical trial white space with the right AI strategy
It has become clear that our industry has reachedthe limits of human-only clinical development. As clinical trials have become increasingly complex, the endeavors that people alone can perform are no longer sufficient to generate the momentum needed to address the growing burden of human disease. This has led to longer drug development timelines and significant delays for patients. One large are of lost time is “white space,” definied simply as unproductive time caused by manual, sequential processes and fragmented data systems. Thankfully, a solution lies in agentic AI and its abilities to perform series of tasks.


More than a tool: How AI agents are transforming clinical trials |
Explore how agentic AI is transforming clinical trials by automating routine tasks and boosting team efficiency with real-world, low-risk use cases.


Why cloud-based eCOA software is becoming the industry standard
The landscape of clinical trials is evolving rapidly, and at the forefront of this transformation is eCOA (electronic Clinical Outcome Assessment) software. This technology is streamlining data collection, enhancing patient compliance, and ensuring regulatory adherence like never before.
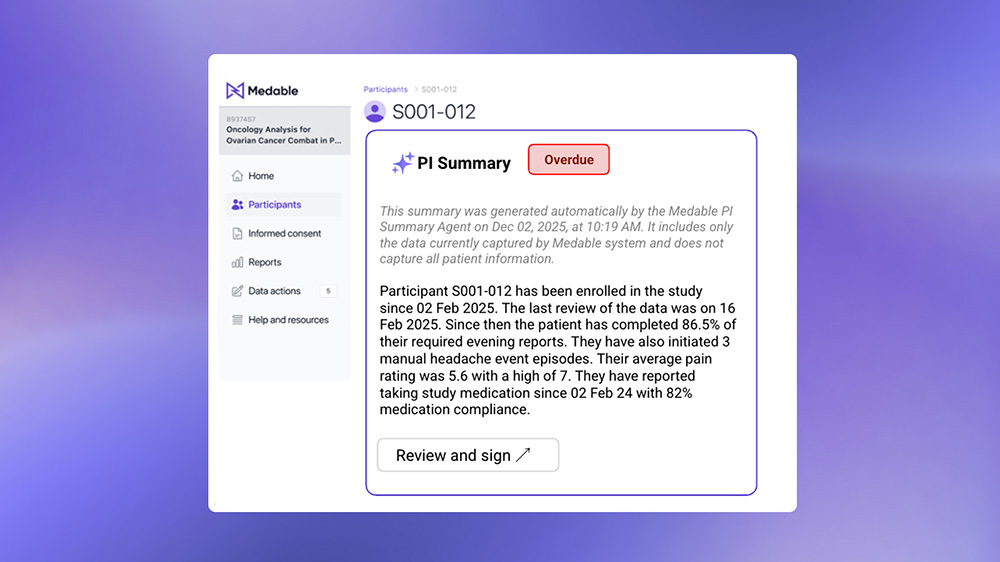

Medable Introduces AI Agent to Reduce Burden at Research Sites by Assisting Principal Investigators with Oversight of eCOA Data
Medable Inc., the leading technology platform for AI-powered clinical development, today announced the launch of Agentic AI for research sites to reduce burden and assist principal investigators in oversight and monitoring of eCOA data.


Medable Debuts AI Agent for Automating Trial Master File (TMF) Processes
TMF Agent significantly improves quality and consistency while drastically reducing the manual effort in document management. It is built on Medable’s Agent Studio, the industry’s first agentic AI platform for clinical development.


Medable Named a Leader Amid Growing eCOA Adoption
Everest Group’s 2025 Life Sciences eCOA PEAK Matrix® Assessment highlights Medable eCOA leadership in ease of use, AI innovation, and patient-centered design


