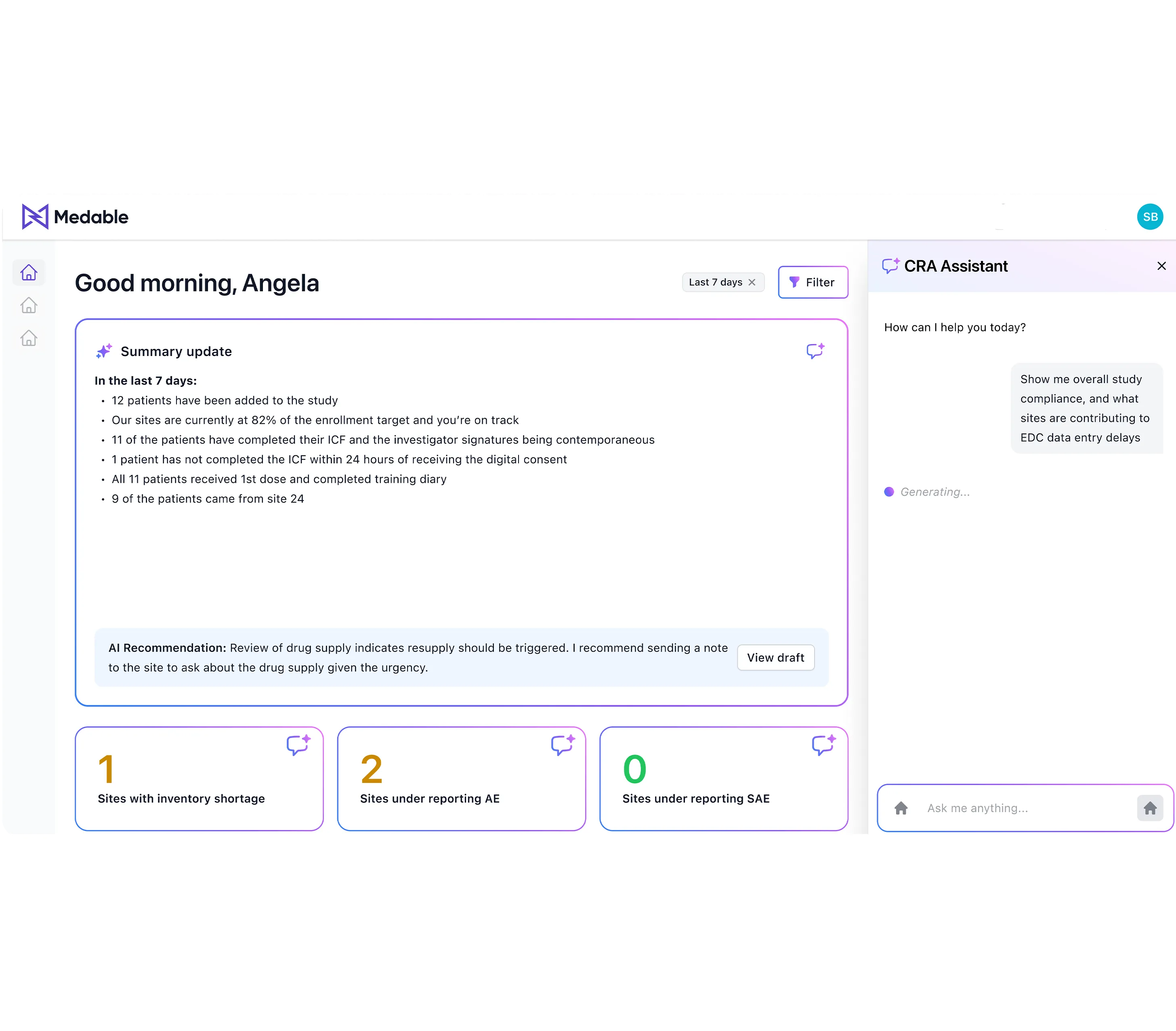
Agentic AI built for clinical development
The only agentic AI platform designed from the ground up for GxP-regulated environments. Model-agnostic. Human-in-the-loop. Connected to 15+ clinical and enterprise systems without replacing any of them.
- Full observability: Transparent, explainable, auditable decision trails
- Connected to your systems: Veeva, Oracle, Medidata, MS 365, Snowflake, and more
- GxP-ready by design: 21 CFR Part 11, Annex 11, ICH GCP, ALCOA+ audit trails
- Human-in-the-loop: Configurable oversight and escalation for every workflow
- Model-agnostic flexibility: Not locked into a single AI provider

Connects to the systems you already use
Over 30 secure, validated connectors to the systems you already run on, from Veeva, Oracle, and Medidata to Snowflake, BigQuery, and your collaboration stack. Your data stays where it lives. Medable just makes it work harder.

The first time I've seen agents in clinical ops this far advanced... you're really ahead of the pack.
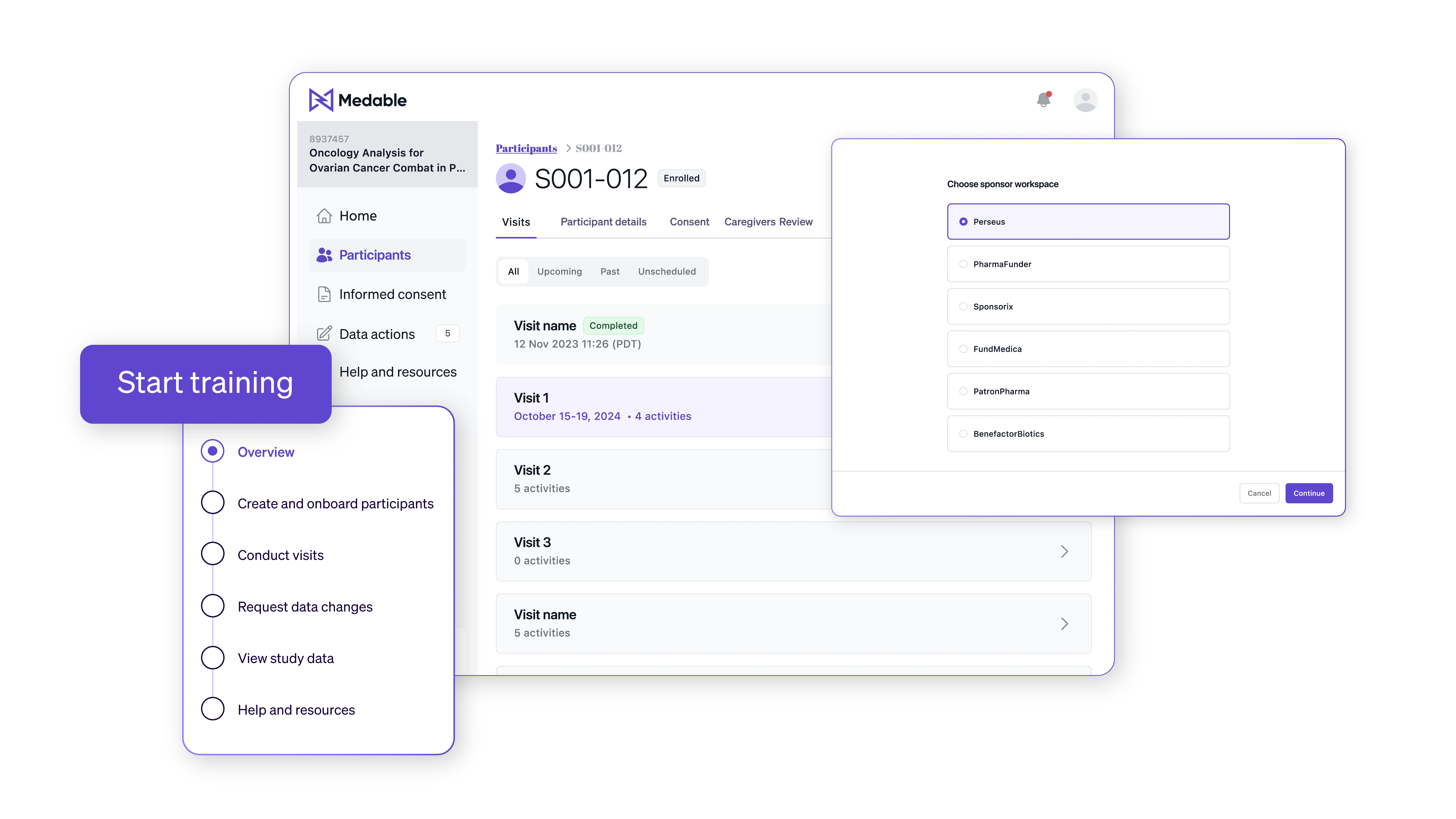
Agentic capabilities across your entire trial
From monitoring to document management to study builds, agentic AI capabilities save you time and protect your data quality.
Platform features
Cloud agnostic API easily integrates data
Learn the fundamental approaches and best practices to create a seamless decentralized approach for clinical trials.
Notifications and reminders
Drive adherence and ensure patients stay engaged with built-in notifications and reminders.
Connectors
MCPs connect systems like EDC, TMF, IRT, and Teams so agents surface insights in Medable Agent Space and take action across your workflows and systems.
Ontology Layer
We normalize your data so agents can deliver insights and act instantly.
Multi-study Participant app
Patients can log into any new study with Medable’s participant app available on both Apple and Google app stores. Study teams mitigate app development and deployment time getting to FPI faster.
24/7 support
Medable offers 24/7 support for trial participants and sites with a 92% first call resolution ensures that help is only a click or call away.
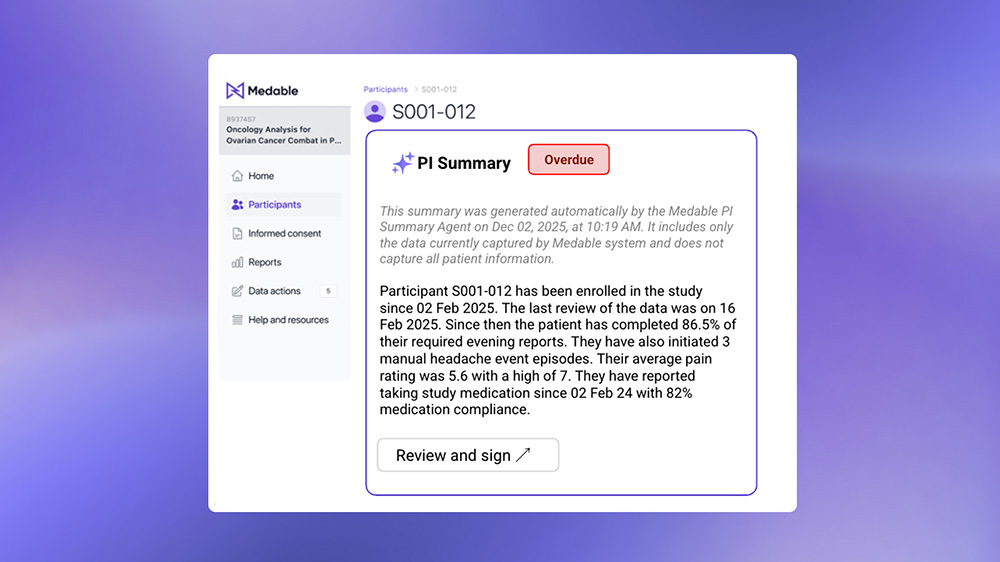
Reporting and dashboards
Gain a quick snapshot of your trial’s status, or deep dive into the nuances of your sites and patients, now supported by Medable AI for faster analysis.
BYOD and Provisioned Devices
Give your sites and patients the convenience and comfort they deserve with BYOD, or our 99% on-time provisioned devices.
What our customers are saying
"Everything has been remarkably smooth, which is not always the case with eCOA vendors."
Top 10 Pharma
"When it comes to modern eCOA solutions Medable is leading the pack."
Director, eClinical Development, Top 5 CRO
"Medable offered robust early engagement and SME dialogue early in planning."
Director, DCT Sourcing, Top 5 Pharma
Platform accolades






Case study: Scaling global vaccine mega-trials for a top-5 pharma
Learn how Medable enabled a top-5 pharma to scale vaccine mega-trials with near-100% enrollment, real-time safety data, and >90% diary compliance.
The latest from Knowledge Centers


The best AI tools for remote patient monitoring in clinical trials
AI-powered remote patient monitoring (RPM) is transforming clinical trials by enabling continuous data collection, real-time insights, and decentralized participation. This ecosystem spans wearables, AI analytics, data platforms, and decentralized clinical trial (DCT) infrastructure.
Additionally, agentic AI is fundamentally reshaping remote patient monitoring (RPM) in clinical trials by shifting it from passive data collection to proactive, autonomous decision support. Instead of simply aggregating data from wearables and patient-reported outcomes, agentic systems can continuously analyze multi-source trial data, identify emerging risks, and take action, such as prioritizing at-risk patients or sites, triggering alerts, or recommending interventions, without waiting for human input. This significantly reduces delays in detecting safety signals or protocol deviations. Just as importantly, agentic AI introduces workflow automation at scale by handling routine monitoring tasks, coordinating communications, and maintaining audit-ready reasoning trails. The result is a more adaptive and responsive RPM model where clinical teams move from manual oversight to strategic supervision, enabling faster, safer, and more efficient trials.
Below is a structured overview of the leading vendors, tools, and providers enabling AI-driven RPM in clinical research.


From three meetings to one removing bottlenecks with AI-enabled eCOA
Discover how AI-enabled eCOA and agentic workflows reduce clinical trial startup time, translation cycles, and meeting overhead—cutting eCOA build timelines from 16–20 weeks to under 8 weeks.


The 1:1:1 vision: Reimagining clinical development
"The scarcest resource in clinical trials is the time of the highly qualified people running the clinical trials. We need to free up their time to bring more meaningful innovation to patients."
- David Hyman, Chief Medical Officer, Eli Lilly
Since the year 2000, the pace of drug approvals has remained stubbornly slow, with the FDA approving roughly 50 new treatments per year. This pace is in spite of massive increases in R&D investment. It’s well known that clinical trials take 10-12 years on average to complete all four phases. But what if clinical trials didn’t take weeks to start, months to enroll, and years to complete?







%20(3).gif)