Clinical development, meet agentic AI.
A new model for clinical development is taking shape — where startup, enrollment, and conduct move in days and months, not years. One platform. Unified workflows, data, and clinical context. Get the full power of agentic AI across the full trial lifecycle at your fingertips




Unlock the agentic advantage
Unify your clinical systems and workflows, and turn data into intelligent, trusted action while reducing manual burden across clinical operations.
Medable’s Agent Platform is purpose-built for clinical development, with deep context across GxP compliance, regulatory requirements, and the operational complexity that general-purpose AI platforms aren't equipped to navigate.
.gif)
Less risk. More control. eCOA, reimagined.
Medable's agentified eCOA brings AI-powered workflows to every stage of the trial — from build and deployment to study conduct and data review — delivering consistent, high-quality data and stronger participant adherence.
Spanning eConsent, sensors, provisioned devices, and BYOD, Medable offers a unified experience that reduces operational complexity across every touchpoint of the trial.
Clinical outcomes backed by real-world evidence
Near-perfect compliance across a 70+ site master protocol.
timelines
Half the time to go-live with AI-powered translation.
From weeks to one business day with Medable Studio.
FPI achieved ahead of schedule across a complex regional rollout.

Trusted, traceable, science-ready AI
Autonomous where it can be, human-led where it must be. Clinical judgment, amplified — never replaced.
- Always traceable: Every automated action logged, explainable, and auditable end-to-end
- Human-in-the-loop: Critical decisions stay with your team giving you full control
- Built-for-purpose: Regulatory context built in from day one — not retrofitted after
Built for global regulatory compliance and data integrity




%20GCP.png)


The 1:1:1 vision
1 day study start, 1 day patient enrollment, 1 year study conduct.
An ambitious vision and one Medable is actively building toward. With agentic AI purpose-built for clinical development, we're making meaningful progress toward timelines the industry once thought were out of reach.

The clinical monitoring agent gives us a holistic view, but what really matters is the site. Normally, we find out about enrollments or screen failures through hundreds of emails, easy to miss. Having that site-specific data right when we log in is a real game changer.
The latest news
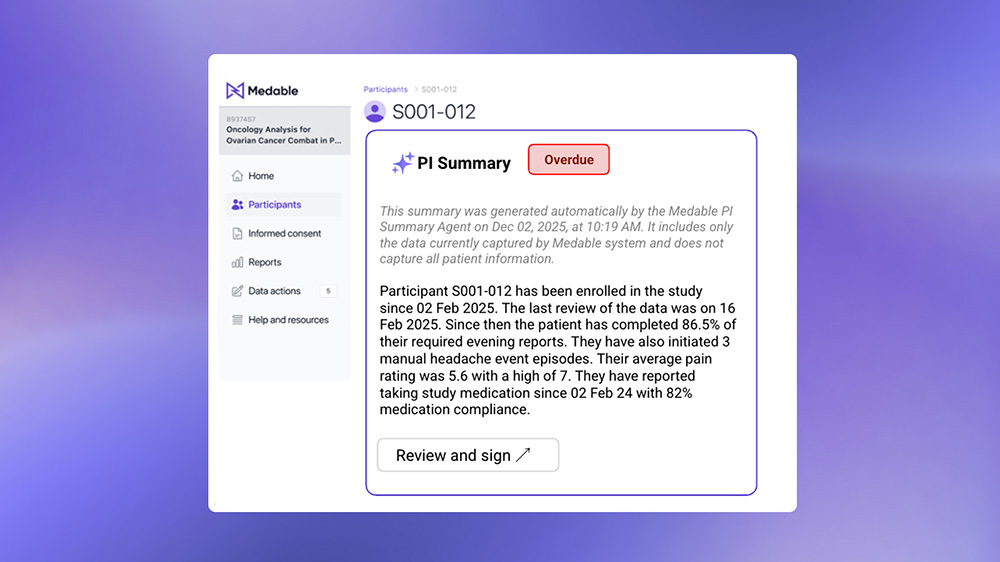

Medable Introduces AI Agent to Reduce Burden at Research Sites by Assisting Principal Investigators with Oversight of eCOA Data
Medable Inc., the leading technology platform for AI-powered clinical development, today announced the launch of Agentic AI for research sites to reduce burden and assist principal investigators in oversight and monitoring of eCOA data.


Medable Debuts AI Agent for Automating Trial Master File (TMF) Processes
TMF Agent significantly improves quality and consistency while drastically reducing the manual effort in document management. It is built on Medable’s Agent Studio, the industry’s first agentic AI platform for clinical development.


Medable Named a Leader Amid Growing eCOA Adoption
Everest Group’s 2025 Life Sciences eCOA PEAK Matrix® Assessment highlights Medable eCOA leadership in ease of use, AI innovation, and patient-centered design




